
The added inner shells are effective at shielding valence electrons which allows for distinct increases in size. The variation in atomic size of the elements in the same group can also be explained by the concept of electron shielding since going down a column increases the principal quantum number n and the number of orbitals between the nucleus and the valence electrons. The effective nuclear charge is greater towards the right side and that is the reason why the atomic radius decreases as we shift from left to right. When studying the periodic table for the MCAT, remember that shifting from left to right across the same period will increase the number of protons in the nucleus while keeping the number of shielding electrons constant since only the number of valence electrons increases across the same period. 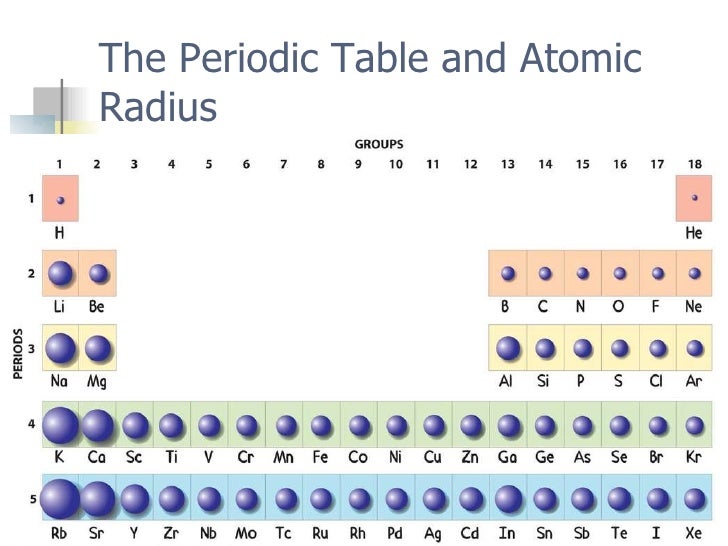
Conversely, the greater the effective nuclear charge, the smaller the atomic radius. Strong “shielding” will not allow the valence electrons to be strongly pulled by the nucleus and they will be free to spread out, thus making the atomic radius bigger. Visualize these shielding electrons as the ones that guard and “shield” the valence electrons from the attractive force of the positive nucleus. Recall that the effective nuclear charge is the net positive charge experienced by valence electrons or the difference between the number of protons and the shielding electrons located in the energy levels between the nucleus and the valence electrons. If you need a more in-depth look at the periodic trends for the MCAT, Blueprint Prep’s Online MCAT Course students can attend Office Hour sessions specifically about this topic.Įven though it is not possible to specify a definite value for the radius of an isolated atom, the atomic radius can be thought of as half the single-bond length of a homonuclear bond and atomic radii variations are closely related to the concept of effective nuclear charge. So we’re focusing on explaining periodic trends and discussing variations in the atomic radius, ionic radius, ionization energy, electron affinity, and electronegativity. Even though the MCAT rarely includes questions asking directly about periodic properties, knowing the periodic trends is fundamental to answering many multiple choice questions in the science sections. An immensely useful tool is the periodic table for the MCAT. Luckily, there are various resources provided during the MCAT exam that can guide you towards the right answers. I can recall how dreadful it was to enter the MCAT testing room with simply an identification card and noise-canceling headphones, questioning whether you have the right tools to tackle the exam or not.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
